C21H40O2
C23H44O2,
C25H48O2
min. 91.0 %
48076-38-6 (C20)
18299-85-9 (C22)
256-350-1 (C20)
242-182-6 (C22)
C21H40O2
C23H44O2,
C25H48O2
min. 91.0 %
48076-38-6 (C20)
18299-85-9 (C22)
256-350-1 (C20)
242-182-6 (C22)
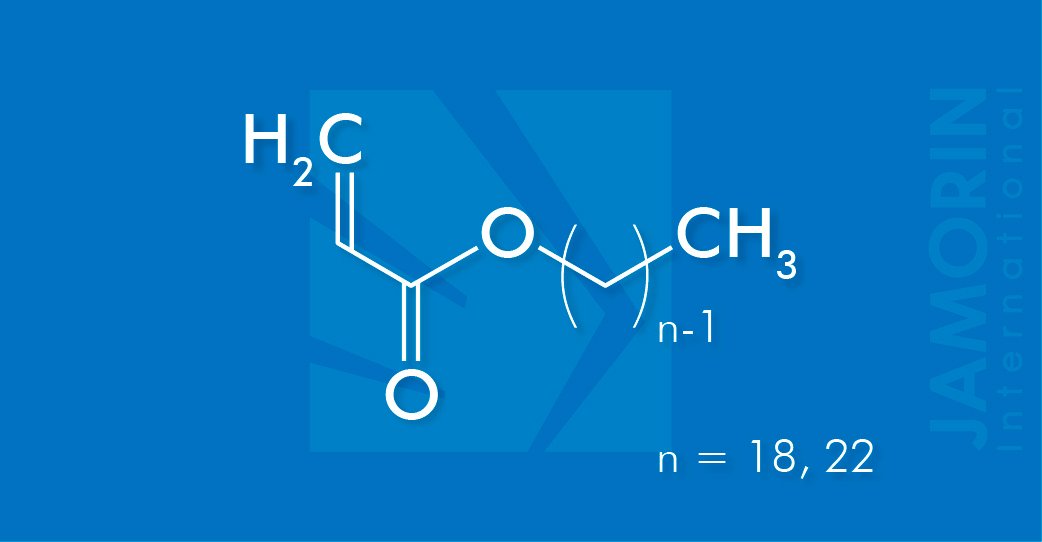
Behenyl Acrylate 1822 (BEA 1822)
Behenyl Acrylate 1822 (BEA 1822) is an ester of Acrylic acid and is used as a raw material component in the synthesis of polymers. Behenyl Acrylate 1822 (BEA 1822) is a monofunctional monomer with a long pendant aliphatic chain and the high reactivity of acrylates. Behenyl Acrylate 1822 (BEA 1822) forms homopolymers and copolymers. The copolymers of Behenyl Acrylate 1822 (BEA 1822) can be prepared with (meth)acrylic acid and its salts, amides, and esters, and with methacrylates, acrylonitrile, maleic acid esters, vinyl acetate, vinyl chloride, vinylidene chloride, styrene, butadiene, unsaturated polyesters, and drying oils, etc.
Behenyl Acrylate 1822 (BEA 1822) is also a very useful feedstock for chemical syntheses because it readily undergoes addition reactions with a wide variety of organic and inorganic compounds.
Product Type: Acrylate monomer
Features & Benefits:
- Chemical stability
- Hydrophobicity
- Abrasion resistance
- Hydrolytic resistance
- Flexibility
- Impact strength
- Low shrinkage
- Weatherability
- Rheology modifier
It is applied in the production of:
- Pour-point depressant
- Caulk, coatings, and inks
- Cosmetics and personal care for skin and hair care
Chemical Properties:
-
Puritymin. 91.0 %
-
Acid Valuemax. 0.05 %
-
Water contentmax. 0.05 %
-
Color APHAmax. 200
Physical Properties:
-
Physical formsolid
-
Molecular weight324.4 g/mol (C18) / 352.6 g/mol (C20) / 380.7 g/mol (C22)
-
Polymer Tg54 (Polymer Tm not Tg)
-
Tgn.a.
-
Density0.8 g/cm3 at 25 °C
-
Boiling Point410 °C
-
Melting Point43 – 45 °C
-
Vapor Pointn.a.
-
Peroxide content (ISO 3960)max. 5 ppm
In order to prevent polymerization, Behenyl Acrylate 1822 F (BEA 1822 F) must always be stored under air, and never under inert gases. The presence of oxygen is required for the stabilizer to function effectively. It has to contain a stabilizer and the storage temperature must not exceed 35 °C. Under these conditions, the storage stability of one year can be expected upon delivery. In order to minimize the likelihood of over storage, the storage procedure should strictly follow the “first-in-first-out” principle. If Behenyl Acrylate 1822 F (BEA 1822 F) is crystallized the product can be melted safely with heating temperatures up to 60 °C. It should not be stored at this temperature for more than 5 days in order to prevent degradation in quality and premature formation of polymer fractions. In order to reduce the thermal stress during the melting process, air convection should be very good. Under such favorable conditions, melting can be achieved within 24 hours. Storage tanks and pipes should be made of stainless steel or aluminum. Storage tanks, pumps, and pipes should be earthed.
Storage & Handling:
In order to prevent polymerization, Behenyl Acrylate 1822 F (BEA 1822 F) must always be stored under air, and never under inert gases. The presence of oxygen is required for the stabilizer to function effectively. It has to contain a stabilizer and the storage temperature must not exceed 35 °C. Under these conditions, the storage stability of one year can be expected upon delivery. In order to minimize the likelihood of over storage, the storage procedure should strictly follow the “first-in-first-out” principle. If Behenyl Acrylate 1822 F (BEA 1822 F) is crystallized the product can be melted safely with heating temperatures up to 60 °C. It should not be stored at this temperature for more than 5 days in order to prevent degradation in quality and premature formation of polymer fractions. In order to reduce the thermal stress during the melting process, air convection should be very good. Under such favorable conditions, melting can be achieved within 24 hours. Storage tanks and pipes should be made of stainless steel or aluminum. Storage tanks, pumps, and pipes should be earthed.
A Safety Data Sheet has been compiled for Behenyl Acrylate 1822 F (BEA 1822 F) that contains up-to-date information on questions relevant to safety.
The data contained in this publication are based on our current knowledge and experience. In view of the many factors that may affect the processing and application of our product, these data do not relieve processors from carrying out their own investigations and tests; neither do these data imply any guarantee of certain properties, nor the suitability of the product for a specific purpose. Any descriptions, drawings, photographs, data, proportions, weights, etc. given herein may change without prior information and do not constitute the agreed contractual quality of the product. It is the responsibility of the recipient of our products to ensure that any proprietary rights and existing laws and legislation are observed.